Reassessing Biosafety risk factors
An interesting assessment of current trends in biosafety, being presented at the WHA:
WHA Provisional agenda item 20. 28 April 2021
A74/18 'Enhancement of laboratory biosafety'
Under 'The way forward' section at the end:
@JamieMetzl
apps.who.int/gb/ebwha/pdf_f… 
This is all too correct.
The technologies mentioned are NGS, which made sequencing faster, easier and so much cheaper but may also mean GoFs, in silico modelling, genome editing, 'de silico' creation, etc. i.e.: the biotechnology revolution toolkit.
mck.co/2QYpGl5 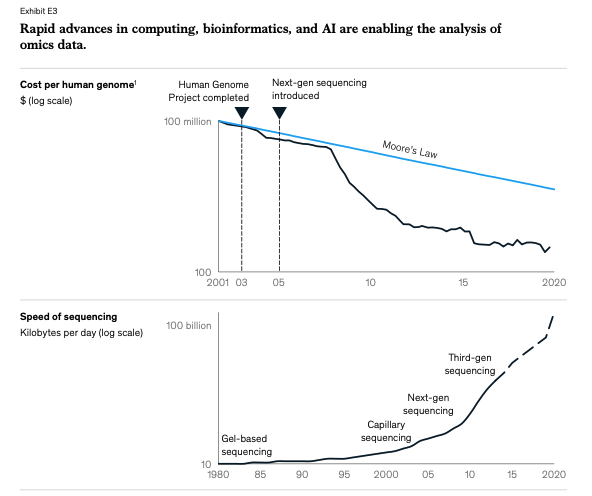
And yes it is an issue that we are still using some dated risk evaluation when we are rushing ahead with all these new tools, deployed in so many new labs.
There is not even a reliable count of P3s and P4s.
The globalbiolabs.org/map is first step for P4s.
@FilippaLentzos
For P3s it is a jungle. In China but also in other countries to be fair.
For instance @RdeMaistre and I did a detailed count of Chinese P3s in China and ended up way above all the official numbers.
And every month that goes by more are coming in service. 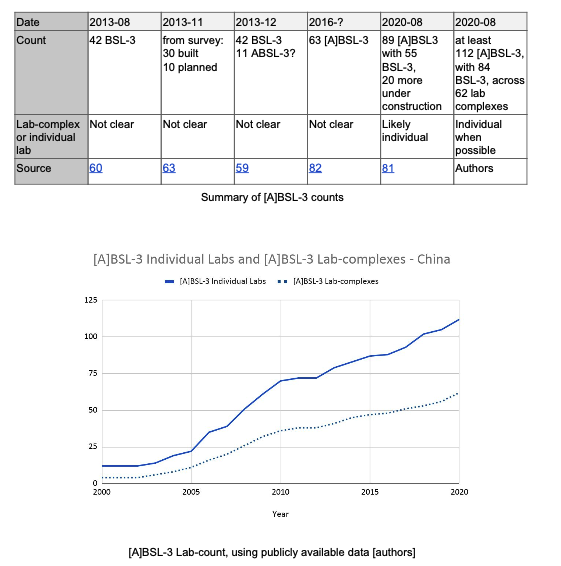
From the Chinese point of view they are just trying to catch with the US, and the faster the better.
Sometimes there is no proper overall plan, infrastructure design, qualified resources, or simply maintenance budget.
gillesdemaneuf.medium.com/evaluation-of-…
The Chinese P3s should thus be a big concern - money and human resources go to the P4s and some key P3s - but they are P3s that have to do with little - especially when built ahead without much consideration.
Annex D6 of the China-WHO report has a good example of that: 
Which is in line with what Yuan Zhiming (manager of the WIV P4) was saying back in October 2019 about P2s and P3s in China: 
@threadreaderapp compile